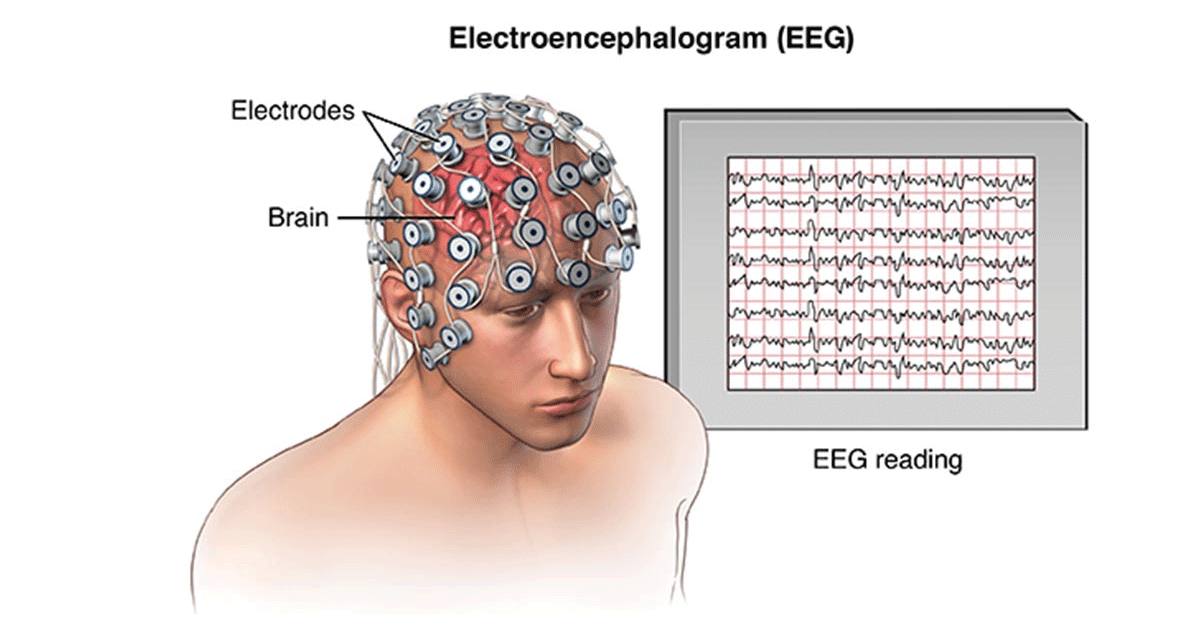
Neurofeedback – also called EEG-Biofeedback – is a scientifically proven brain training to improve certain functions of the brain. Patients learn to change their brain waves to enhance certain skills, such as concentration, memory, top performance, creativity, relaxation, etc. Since more than 15 years the German Neuroscience Center is the leading clinic for Neurology, Psychiatry and Psychology in the UAE. We are offering the highest quality Neurofeedback treatment with the leading Neurofeedback experts in the region.
Which Disorders Are Treated With Neurofeedback?
Neurofeedback is a painless and non-invasive procedure that has been used for a variety of diseases for over 30 years.
- ADHD, ADD – Attention Deficit (Hyperactivity) Disorders (Evidence base level 5)
- Epilepsy (Evidence base level 5)
- Learning disabilities
- Autism and Asperger Syndrome
- Depression
- Anxiety and panic disorders, post-traumatic stress disorders, obsessive compulsive disorders, sleep disorders, chronic fatigue syndrome, traumatic brain injuries, fibromyalgia and chronic pain syndromes, migraines, autism, dementia, tic disorders as Tourette Syndrome, Parkinson Disease, Lyme disease and can be treated as well.
Neurofeedback is a new treatment option for the above-mentioned disorders to achieve rapid and permanent changes in the areas of cognition, behavioral problems, affective regulation, and social adaptation and function.
What Does A Neurofeedback Session Look Like?
The patient is sitting relaxed in front of a screen. The activity of the brain waves is recorded by means of sensors (EEG recording) and displayed graphically by a computer program. The patient is given various tasks in order to change the brain waves. The brain receives information about its activity and learns to optimize it by means of feedback that is perceived via images, videos, sound etc.
How Does Neurofeedback Work?
The patient’s brain waves are recorded using an EEG. A special computer program divides the brain waves into frequency ranges and analysis them. Depending on the clinical picture, certain pathological patterns of brain activity are recognized, and the patient is given appropriate feedback. There are many different forms of feedback such as films, tones, games, etc. The patient should learn to encourage certain activities or suppress others. The patient uses this to train his brain until the desired pattern occurs and the symptoms are improved.
The feedback is based on the principle of so-called operant conditioning. This means: The feedback given to the patient is a reward if the patient succeeds in activating certain areas of the brain in such a way that they generate the desired brain activity. Our brain wants to be rewarded as often as possible. It tries to permanently recognize and understand the patterns hidden in the feedback in order to receive a reward as often as possible. This process happens completely unconsciously, without active intervention by the patient and without medication.
What Are Brain Waves?
In a raw EEG brain activity is divided by frequencies: GAMMA greater than 30(Hz) BETA (13-30Hz), ALPHA (8-12 Hz), THETA (4-8 Hz), and DELTA (less than 4 Hz). Faster waves are needed to complete a complex task, slower waves are seen when relaxing or sleeping. However, brain waves always appear as a mix. Imbalances can cause disorders such as being nervous or anxious when too many fast waves are seen in the EEG.